 
I put g/mol for the unit because that what was used in the problem statement.Įxample #6: A sample of element X contains 100 atoms with a mass of 12.00 and 10 atoms with a mass of 14.00. Calculate the average atomic mass of lithium.ġ) Calculate the percent abundance for each isotope: 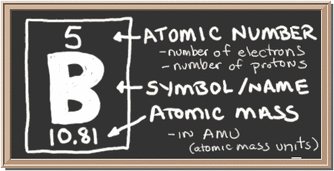
(2) I forgot to put a unit on the answer, so 186.207 u would be the most correct answer.Įxample #5: In a sample of 400 lithium atoms, it is found that 30 atoms are lithium-6 (6.015 g/mol) and 370 atoms are lithium-7 (7.016 g/mol). Solution: Two points: (1) notice I wrote the same number of decimal places in the answer as were in the isotopic weights (the 184.953 and the 186.956). To obtain the correct answer, we must divide by 100.Įxample #4: How to Calculate an Average Atomic Weight Our answer is too large by a factor of 100. When discussing the atomic weight of an isotope, this value is a value that has been measured experimentally, not an average.Įxample #3: Silicon mass number isotopic weight percent abundance 
(isotopic weight) (abundance) + (isotopic weight) (abundance) = average atomic weightĪ point about the term 'atomic weight:' When discussing the atomic weight of an element, this value is an average. Consult a periodic table to see what manner of answers are considered acceptable.Įxample #2: Nitrogen mass number isotopic weight percent abundance The answers to problems like this tend to not follow strict significant figure rules. This is commonly rounded to 12.011 or sometimes 12.01. Then, add the results together and round off to an appropriate number of significant figures. Solution: To calculate the average atomic weight, each isotopic atomic weight is multiplied by its percent abundance ( expressed as a decimal). Mass number isotopic weight percent abundance Study the tutorial below and then look at the tutorial linked just above.Įxample #1: Calculate the average atomic weight for carbon. This problem can also be reversed, as in having to calculate the isotopic abundances when given the atomic weight and isotopic weights. The older symbol (which the ChemTeam grew up with) is amu (sometimes seen as a.m.u.) The unit amu is still in use, but you will see u used more often. If it is not clear from the context that g/mol is the desired answer, go with amu (which means atomic mass unit).īy the way, the most correct symbol for the atomic mass unit is u. The unit associated with the answers to the problems below can be either amu or g/mol, depending on the context of the question. The ChemTeam prefers to use Wikipedia to look up values. These values can be looked up in a standard reference book such as the "Handbook of Chemistry and Physics." The values can also be looked up via many online sources. (b) the percent abundance for each isotope (a) the exact atomic weight for each naturally- occuring stable isotope To do these problems you need some information. ChemTeam: Calculate the average atomic weight from isotopic weights and abundancesĬalculate the average atomic weight when given isotopic weights and abundancesĬalculate the isotopic abundances, given the atomic weight and isotopic weights 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
